Ph Eur
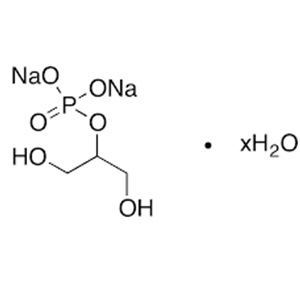
C3H7Na2O6P, xH2O -- 216.0 (anhydrous substance)
DEFINITION
Mixture of variable proportions of sodium (2RS)-2,3-dihydroxypropyl phosphate and sodium 2-hydroxy-1-(hydroxymethyl)ethyl phosphate. The degree of hydration is 4 to 6.
Content: 98.0 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance: White or almost white, crystalline powder or crystals.
Solubility: Freely soluble in water, practically insoluble in acetone and in alcohol.
IDENTIFICATION
A. Solution S (see Tests) gives reaction (a) of sodium.
B. To 0.1 g add 5 ml of dilute nitric acid R. Heat to boiling and boil for 1 min. Cool. The solution gives reaction (b) of phosphates.
C. In a test-tube fitted with a glass tube, mix 0.1 g with 5 g of potassium hydrogen sulphate. Heat strongly and direct the white vapour into 5 ml of decolorised fuchsin solution. A violet-red colour develops which becomes violet upon heating for 30 min on a water-bath.
TESTS
Solution S: Dissolve 10.0 g in carbon dioxide-free water prepared from distilled water and dilute to 100 ml with the same solvent.
Appearance of solution: Solution S is not more opalescent than reference suspension II (and not more intensely coloured than reference solution.
Alkalinity: To 10 ml of solution S add 0.2 ml of phph solution. Not more than 1.0 ml of 0.1 M hydrochloric acid is required to change the colour of the indicator.
Glycerol and alcohol-soluble substances: Maximum 1.0 per cent.
Shake 1.000 g with 25 ml of alcohol R for 10 min. Filter. Evaporate the filtrate on a water-bath and dry the residue at 70 °C for 1 h. The residue weighs not more than 10 mg.
Chlorides: Maximum 200 ppm.
Phosphates: Maximum 0.1 per cent.
Sulphates: Maximum 500 ppm.
Iron: Maximum 20 ppm.
Heavy metals: Maximum 20 ppm.
Water: 25.0 per cent to 35.0 per cent, determined on 0.100 g.
ASSAY
Dissolve 0.250 g in 30 ml of water. Titrate with 0.05 M sulphuric acid , determining the end-point potentiometrically.
Calculate the percentage content of sodium glycerophosphate (anhydrous substance) from the expression:
216(n1-n2/4)
m(100-a)